How the easy-HC10 handheld disinfectant maker and sprayer works
Disinfectant fogger. What is hypochlorous acid?
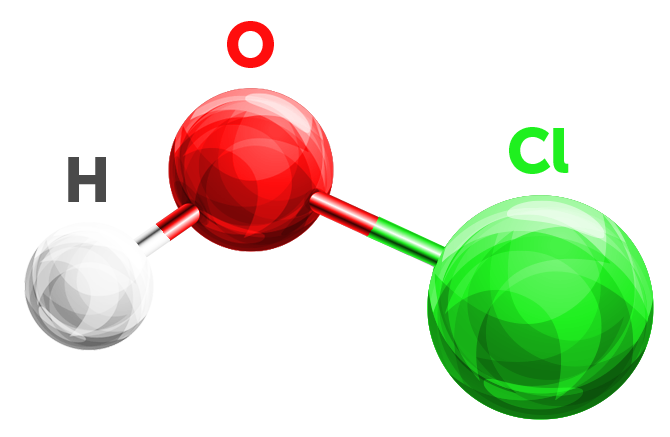
Hypochlorous acid (HClO or HOCl) is a weak acid that forms when chlorine dissolves in water and partly dissociates forming hypochlorite (ClO⁻). HClO and ClO⁻ are oxidizers and the primary disinfecting agents of chlorine solutions.
When dissolved in water hypochlorous acid has been found to have very effective disinfection properties; this, in conjunction with its economical (just electrolysed salt and water) and non-toxic characteristics, makes it an ideal disinfectant fogger, cleaning agent and sanitizer. [2] Many products with hypochlorous acid as the disinfectant are listed on the US Environmental Protection Agencies list N: Disinfectants for Coronavirus (COVID-19).
The easy-HC10 disinfectant fogger has successfully exceeded the stringent criteria of disinfectant and biocide testing undertaken by an independent UKAS accredited laboratory. These tests evaluated the quality of the hypochlorous acid produced in-situ to kill coronavirus (BS EN 14476:2013+A2:2019), bacteria (BS EN 1276:2019), and yeast (BS EN 1650:2019):
- Virucidal test against coronavirus with just 45 ppm (under BS EN 14476:2013+A2:2019)
- Bactericidal properties (Four different bacteria under test BS EN 1276:2019)
- Yeasticidal properties (under test BS EN 1650:2019)
How the easy-HC10 works
Disinfectant fogger, start by filling the 2 litre reservoir with tap water. Then add 5 grams of sodium chloride (salt) and give it a shake. The salt solution is pumped from the reservoir into the micro-IonTM continuous flow electrochemical reactor.
This micro-IonTM chemical reactor is integrated into the portable handheld sprayer. Within the micro-IonTM, Electricity from the battery is conducted through the salt solution using specially made electrodes to oxidise the salt solution favouring free cholrine to produce hypochlorous acid.
The hypochlorous acid is then pumped from the micro-IonTM to the nozzle where it is atomised into a mist and carried to the surface to be disinfected by a high speed jet of air. The disinfectant is made within the micro-IonTM reactor only 1 to 2 seconds before spraying.
There is no need to worry about degradation or shelf life of pre-purchased disinfectants. The whole process is all completed in continuous flow as soon as the trigger is pressed. There is no waiting necessary for the disinfectant to generate.

How is hypochlorous acid so effective against pathogens?
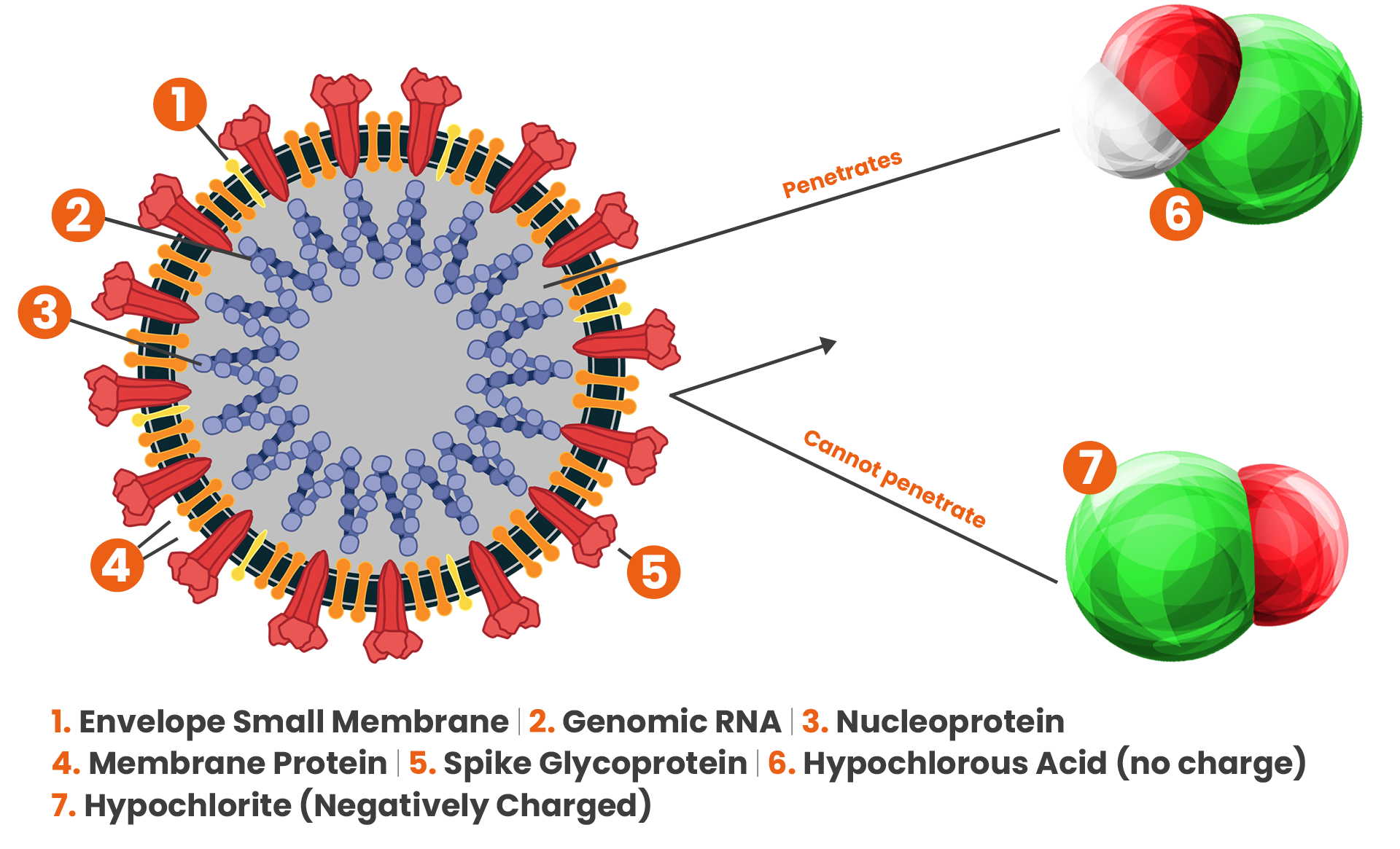
As chlorine dissolves in water, it partly dissociates forming hypochlorite (ClO⁻). Hypochlorite is also known as bleach and is an effective disinfectant but not as effective as hypochlorous acid. This is because hypochlorite is negatively charged and so are bacteria.
This causes them to act like a magnet if you put them together the wrong way around, meaning they repel each other and therefore reduces the overall effectiveness of hypochlorite. As hypochlorous acid is not charged at all, it is not repelled by bacteria. This means it can easily penetrate the protective walls of bacteria and render it safe.
The pH of the hypochlorous acid produced by the micro-IonTM is maintained in the range pH 5.5 – pH 7. pH plays a big role in the effectiveness of hypochlorous acid as a disinfectant, this is because at pH levels of above 7.5 there will be more than 50% of the disinfectant being hypochlorite (ClO⁻) which is not as effective as hypochlorous acid.
At pH levels of 5.5 to 7.5 the dominating disinfectant will be hypochlorous acid with there being more than 80% of the disinfectant being hypochlorous acid at a pH of 6.5.
About hypochlorous acid
Hypochlorous acid is produced by white blood cells in all mammals. It is an essential part of our immune system as it eliminates pathogens and is also harmless to mammalian cells in low concentrations. [4]
Hypochlorous acid can also be produced through a process called electrolysis. This method of hypochlorous acid production is replicated by the easy-HC10.
Electrolysis is the passing of current through a saltwater solution. With the easy-HC10 it causes a chemical reaction between the specially designed electrodes which, in turn, produces hypochlorous acid. Although hypochlorous acid is a weak acid, it is considered to be a strong oxidising agent that kills a variety of pathogens.
The safety of hypochlorous acid
Hypochlorous acid does not irritate eyes or skin as it is already made naturally in the human body. Hypochlorous acid is also safe to use on food and food surfaces as it eliminates bacteria, fungi, yeasts, viruses, molds and spores.
Also, in early 2016 the US Food & Drug Administration (FDA) approved products whose main ingredient is hypochlorous acid for use in treating certain wounds and infections in humans and pets. The Environmental Protection Agency in the US classified hypochlorous acid as non-hazardous.
Further to this, in a recent clinical test of hypochlorous acid for eye irritation, skin irritation and toxicity, it was concluded that hypochlorous acid is non-irritating to eyes and skin as well as being non-toxic.
The broad uses of hypochlorous acid
- In healthcare environments such as doctors’ surgeries and hospitals hypochlorous acid has been used as a disinfectant and sanitizer. [6][7]
- As of early 2016 the US Food and Drug Administration approved products that have hypochlorous acid as the main ingredient for use in treating wounds and infection in humans and pets. It is also approved by the FDA as a preservative for saline solutions. [1][7]
- Hypochlorous acid is used as a disinfectant in a sprayed fog or mist, wet wipes, and aerosol applications. Recent studies have shown that using hypochlorous acid in this form is suitable in settings such as offices, schools, hospitals, buses, trains, and planes. [9]
- In water treatment hypochlorous acid is the active sanitiser in hypochlorite-based products (e.g., swimming pools). [1]
- In ships and yachts marine sanitation devices are used to convert salt water into hypochlorous acid disinfectant to be used for general cleaning and macerated faecal waste before it is ejected into the sea. [10]
- Hypochlorous acid is also a very effective deodorizer. It has been tested to remove up to 99% of odours which make it perfect for use in toilets, rubbish bins or disposal plants, changing rooms ETC. [11]
References
- https://en.wikipedia.org/wiki/Hypochlorous_acid
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7315945/
- https://cfpub.epa.gov/giwiz/disinfectants/index.cfm
- https://hocl.com/about
- https://en.wikipedia.org/wiki/Electrolysis
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1853323/
- https://www.todayswoundclinic.com/articles/treating-chronic-wounds-hypochlorous-acid-disrupts-biofilm
- https://www.magonlinelibrary.com/doi/abs/10.12968/jowc.2006.15.1.26861
- https://www.dovepress.com/the-potential-use-of-hypochlorous-acid-and-a-smart-prefabricated-sanit-peer-reviewed-article-RMHP
- https://www.maritime-executive.com/index.php/features/new-sanitizing-technology-makes-quick-work-of-covid-19-1
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6922465/
- https://www.cleanroomtechnology.com/news/article_page/The_science_of_chlorine-based_disinfectant/93824

